 Accrediting agencies look for traceability processes that are compliant with evidence-based standards and guidelines. trophon2 with AcuTrace® RFID technology simplifies the creation of accurate digital records, easily obtained from your trophon2 device to support audit-readiness.
Accrediting agencies look for traceability processes that are compliant with evidence-based standards and guidelines. trophon2 with AcuTrace® RFID technology simplifies the creation of accurate digital records, easily obtained from your trophon2 device to support audit-readiness.
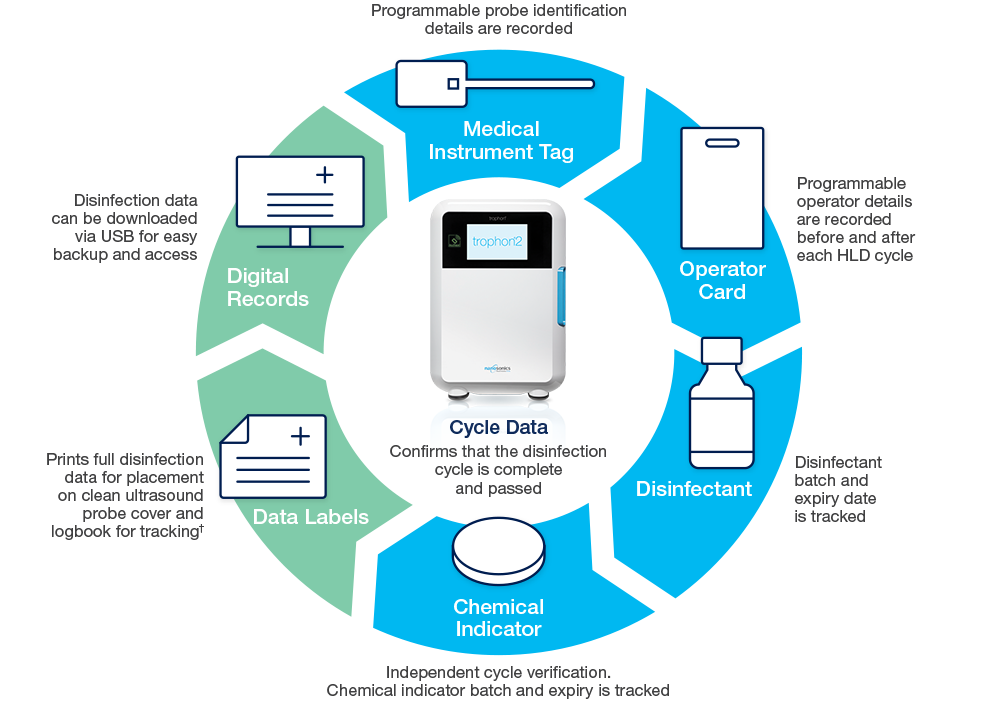
Automated Digital Traceability Across the Workflow
The trophon2 device increases user compliance and supports audit-readiness through automation, capturing data across the entire reprocessing workflow.
AcuTrace RFID technology records operator, probe and cycle data to capture and demonstrate user compliance.
AcuTrace-enabled consumables and accessories include:

Audit-Ready Compliance and Archiving
AcuTrace technology simplifies the creation of accurate digital records, accessible on your trophon2 device to support audit-readiness. AcuTrace technology electronically captures disinfection data in accordance with requirements in USA, CAN, ANZ, UK & EU.1-14
This data is easily accessible to suit a variety of user traceability methods:
- Printer label capturing AcuTrace inputs and disinfection results
- Download disinfection records to USB
- Optional AcuTrace® PLUS allowing API access, supporting digital disinfection data archiving*.

*Custom API middleware required to enable this capability. All connectivity, configuration and integration with customer IT systems is the responsibility of the customer.
The trophon technology family includes trophonEPR devices and trophon2 devices which share the same core technology of 'sonically activated' hydrogen peroxide.
- AORN 2018. High-Level Disinfection. In: AORN Guidelines for periOperative Practice. Denver, CO.
- ANSI/AAMI ST58:2013 Chemical sterilization and high-level disinfection in health care facilities.
- CDC 2008 Guideline for Disinfection and Sterilization in Healthcare Facilities.
- AS/NZS 4815:2006 Office-based health care facilities - Reprocessing of reusable medical and surgical instruments and equipment, and maintenance of the associated environment.
- AS/NZS 4187:2014 Cleaning, disinfecting and sterilizing reusable medical and surgical instruments and equipment, and maintenance of associated environments in health care facilities.
- ACIPC/ASUM 2017. Guidelines for Reprocessing Ultrasound Transducers. Australasian Journal of Ultrasound in Medicine. 2017;20(1):30-40.
- Australian Commission on Safety and Quality in Health Care. National Safety and Quality Health Service Standards, second edition. November 2017.
- CAN/CSA-Z314-18. Canadian Medical Device Reprocessing. February 2018.
- Health Service Executive. Quality Improvement Division (2017). HSE Guidance for Decontamination of Semi‐critical Ultrasound Probes; Semi‐invasive and Non‐invasive Ultrasound Probes.
- Health Protection Scotland (2016). NHSScotland Guidance for Decontamination of Semi-Critical Ultrasound Probes; Semi-invasive and Non-invasive Ultrasound Probes.
- Welsh Health Technical Memorandum (2014). WHTM 01-06 - Decontamination of flexible endoscopes Part C: Operational management,
- Department of Health (UK) (2016). Health Technical Memorandum 01-06 Part C Operational management.
- Ministère des affaires sociales et de la santé (2016). INSTRUCTION N° DGOS/PF2/DGS/VSS1/2016/220 du 4 juillet 2016 relative à relative au traitement des endoscopes souples thermosensibles à canaux au sein des lieux de soins.
- Kommission für Krankenhaushygiene und Infektionsprävention and Bundesinstitut für Arzneimittel und Medizinprodukte. Anforderungen an die Hygiene bei der Aufbereitung von Medizinprodukten

